[ad_1]
Impact of irradiation dose and SBR content material because the compatibilizer of PVC and LLDPE
This text investigated the SBR affect and gamma irradiation doses on the compatibility of PVC and LLDPE compounds. Irradiations of immiscible mix polymers are primarily compatibilized after irradiation21. Quite a few research have proven that γ-rays penetrate deeper and produce radicals that set off cross-linked processes. In contrast with E-Beam radiation, γ-ray has greater penetration energy22,23,24,25. The adverse cost particles of E-Beam radiation restrict its penetration energy26,27,28,29,30.
Morphological Properties of irradiated (PVC/LLDPE)ZnO blends
Determine 1 shows how the gamma radiation doses and SBR content material have an effect on the compatibility of PVC and LLDPE. Differing SBR compatibilizer concentrations produce utterly distinct morphologies. And not using a compatibilizer, the combination displays a dispersed PVC and LDPE section with variable styles and sizes; even a large portion of the domains resemble droplets. SBR compatibilizers resulted in PVC domains with frequent situations and primarily constant measures. The compatibilizers employed have a major impression on the PVC area sizes. It’s typically acknowledged {that a} compatibilizer performs two important roles in managing a mix’s morphology: coalescence prevention and interfacial rigidity discount. As a result of compatibilizers’ position in steric stability, it’s assumed that the homogeneity of the PVC domains’ dimension and form generated by their addition outcomes from a lower in coalescence. The clean samples (0 kGy) comprise extra droplet matrix with extra cavities, as seen in Fig. 1. The blends’ SEM evaluation confirmed that the SBR compatibilizer’s addition and the irradiation technique produced finer mix morphologies with much less roughness. In PVC/LLDPE blends, the SBR compatibilizer lowered droplet coalescence and aided in stabilising wonderful morphology. The compatibility between LLDPE and PVC matrixes was considerably improved by the SBR compatibilizer stage of three wt%. Moreover, gamma irradiation barely improves compatibility quite than simply the SBR impression. The coalescence of lately produced droplets turns into extra important when SBR focus will increase.
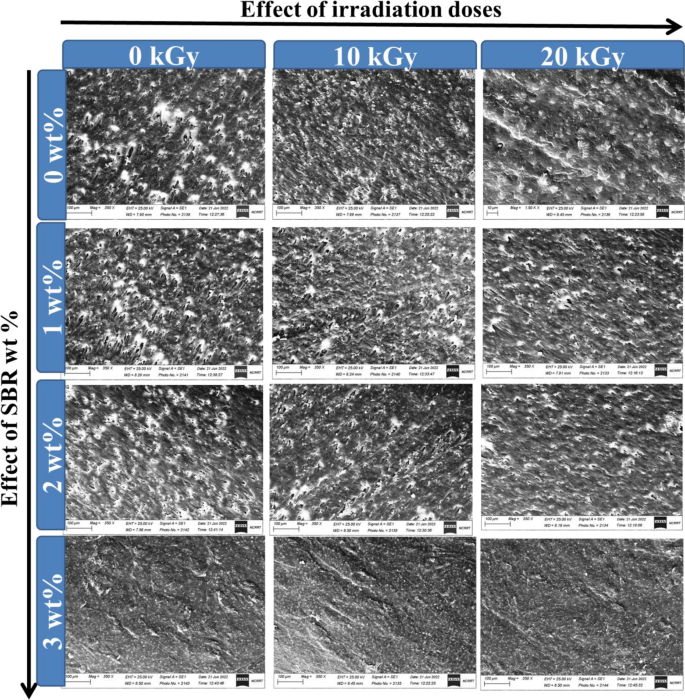
The impact of irradiation doses and SBR content material within the floor morphological of (PVC/LLDPE)ZnO blends.
Mechanical properties of irradiated (PVC/LLDPE)ZnO blends
As a consequence of their low interphase adhesion and excessive interfacial rigidity, PVC and LLDPE type incompatible combos, as reported in a number of tutorial publications31,32,33. The mechanical properties of PVC and LLDPE are virtually inferior33 and might be improved by the SBR addition and gamma irradiation course of. Determine 2 demonstrates how the SBR and irradiation processes enhance the blends’ tensile power and elongation. This result’s as a result of elevated compatibility of PVC with LLDPE. Primarily based on ASTM requirements, a stress take a look at was plotted in Fig. 2a on completely different contents of SBR compatibilizer and gamma irradiation doses. The pressure power of (PVC/LLDPE)ZnO samples is enhanced after including SBR and publicity to gamma radiation than clean samples. The compatibilizer (SBR) has a very good impact on interfacial bonding after the irradiation course of and will increase the pressure power of the (PVC/LLDPE)ZnO blends. The addition of SBR will increase the pressure power and displays superior materials power after gamma irradiation attributable to gamma irradiation-induced crosslinking of SBR34. In Fig. 2b the elongation (mm) is improve about 21%, 30% and 52% with SBR content material improve from 1%, 2% and three% at 0 kGy, respectively. This distinction is as a result of SBR chains made plasticizer impact in blends pattern. Primarily based on the definition of plasticization, the elongation ought to improve with a rise within the plasticizer focus35,36,37,38. After the gamma irradiation course of, the elongation is decreased. For instance, at 3% SBR the elongation decreased from 11 to 16.8% for irradiation doses at 10 kGy and 20 kGy, respectively. This as a result of gamma irradiation induced additional cross-linked reactions and so decreased the motion of chains and elongation39,40.
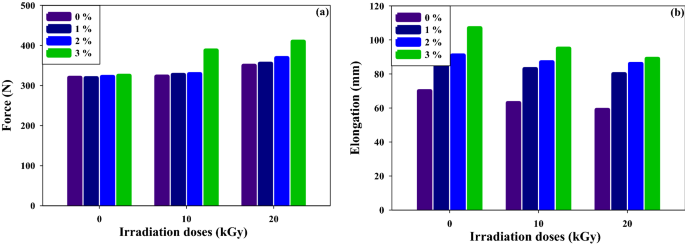
The impact of SBR content material and irradiation doses on the mechanical properties of (PVC/LLDPE)ZnO mix.
Amine-functionalized of PVC in irradiated (PVC/LLDPE)/ZnO mix
Poly(vinyl chloride) (PVC) was chemically modified with 4 completely different amino compounds, together with ethylene diamine (EDA), aniline (An), p-anisidine (pA) and dimethyl aniline (DMA) for bettering the electrical conductivity and oil elimination functionality of the mix polymer. The chemical construction and the proposed mechanism of PVC amination are represented in Fig. 3. After amination reactions, all samples of modified (PVC/LLDPE)ZnO-b exhibit coloration diploma various from brown to darkish brown. All ionomers had been ready by nucleophilic substitution in a solvent/non-solvent system at delicate situations (Desk 1). Chemical modification of PVC by nucleophiles was carried out in a 100-ml three-neck round-bottomed flask outfitted with a magnetic stirrer and condenser. Virtually, 90 v% of ethylene diamine (EDA) dissolved in ethanol was added dropwise to five g (PVC/LLDPE)/ZnO strip soaked in 20 ml of ethanol options. The combination was heated to 80 °C for 3 h after which the modified (PVC/LLDPE)/ZnO strip was immersed in a mixture of ice and water on the thermal equilibrium. The brand new modified (PVC/LLDPE)/ZnO-EDA was obtained and saved in dry situations for additional experimental research. A combination of 5 g of (PVC/LLDPE)/ZnO and 10 ml of aniline dissolved in10 ml of ethanol solvent was stirred for about an hour and at a temperature of 90 °C. After that, the aniline modified (PVC/LLDPE)/ZnO was washed with a combination of ethanol/water. For the modified (PVC/LLDPE)/ZnO with p-Anisidine, a response combination of 5 g and 10 g, respectively, was continued by stirring in 10 ml of ethanol for about 12 h at room temperature. The ensuing modified strip polymer (PVC/LLDPE)/ZnO-pA was washed and dried. A combination of dimethyl aniline (30 ml) dissolved in 10 ml of ethanol containing 5 gm of (PVC/LLDPE)/ZnO strip. The combination was refluxed in 100-ml three-neck round-bottomed flask for 4 h at a temperature of 80 °C. The obtained (PVC/LLDPE)/ZnO-DMA was washed a number of instances with an ethanol/water combination. Determine 3 exhibits the proposed mechanism reactions of Poly(vinyl chloride) (PVC) with 4 completely different amino compounds, together with ethylene diamine (EDA), aniline (An), p-anisidine (pA) and dimethyl aniline (DMA). The functionalized amine PVC was fashioned by the nucleophilic assault of the (N) atom on the carbon-bearing chlorine atom within the polymeric chain of PVC, beginning the method. The chloride anion was left as a very good leaving group (-HCl).
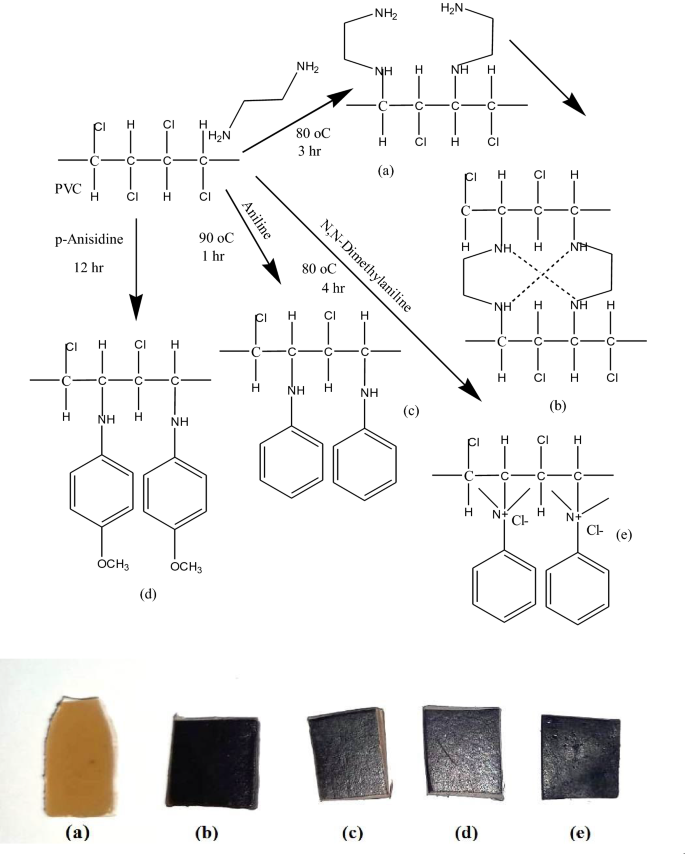
proposed amine-functionalized poly (vinylchloride/Linear low-density polyethylene)/ZnO with the chosen completely different 4 amines; (a) ethylene diamine EDA, (c) aniline An, (d) p-anisidine pA, (e) N, N dimethyl aniline DMA and (b) a attainable crosslinking mechanism for 2 merchandise functionalized by diamine precursors. The photograph in backside is present the altering of samples shade after amination response (a) (PVC/LLDPE)ZnO-b, (b) (PVC/LLDPE)ZnO-EDA, (c) (PVC/LLDPE)ZnO-An (d) (PVC/LLDPE)ZnO-pA and (e) (PVC/LLDPE)ZnO-DMA.
Chemical modification carried out by FTIR/ATR
After the facile chemical modification by amination teams of PVC molecules in (PVC/LLDPE)ZnO-b by way of the nucleophilic chemical reactions. Determine 4 exhibits the FTIR curves of a brand new amine functionalized PVC group fashioned on the (PVC/LLDPE)ZnO-b samples. The chemical construction of the modified (PVC/LLDPE)ZnO was carried out by FTIR, and every of the modified (PVC/LLDPE)ZnO confirmed its attribute FTIR peaks in line with its chemical construction. The profitable modification of PVC with 4 completely different amines might be demonstrated by FTIR spectroscopy in Fig. 4. As proven in Fig. 4a (PVC/LLDPE)ZnO-b cross-linked reactions induced by gamma irradiations happen primarily as a result of radical formations and elimination of chloride ions (e.g., –HCl)41. This dechlorination course of is accountable for forming brief polymeric chains containing C=O and –CH=CH– moieties. As irradiation of (PVC/LLDPE)ZnO-b progresses, the formation of the C=O and –CH=CH– turns into noticeably important. Subsequently, FTIR spectroscopy was used to look at the expansion of the absorption peaks akin to the C=O and –CH=CH– teams positioned at 1651 cm-1 and 1602 cm-1, respectively. The adjustments within the place of C=O and C=C for the (PVC/LLDPE)ZnO after chemical modification are predicted as proven in Figs. 4b-e. It was clear that the adjustments within the C=O and C=C had been considerably extra important and sharper within the case of the (PVC/LLDPE)ZnO-b. As well as, the 2 FTIR peaks at 2913 cm-1 and 2884 cm-1 correspond to the uneven and symmetric stretching vibration of –CH– is a repeat unit in LLDPE and PVC molecules. The FTIR peaks at 722 cm-1 and 1462 cm-1 correspond to the stretching vibration of C–Cl and the bending vibration of –CH2 in PVC and LLDPE molecules, respectively. Determine 4b exhibits the FTIR curve of (PVC/LLDPE)ZnO–EDA pattern, the attribute two peaks positioned at 1566 cm-1 and 820 cm-1 are assigned to bending vibration of (N–H) and (C–N) bonds, respectively. The height positioned at 1330 cm-1 corresponds to the stretching vibration of (C–N) bonds in PVC/LLDPE)ZnO-EDA pattern. As well as, the broad beak positioned at 3410 cm-1 corresponds to the starching vibration of (N–H) in main amine. The broadening peak of 3410 cm-1 is as a result of intramolecular hydrogen bonds excited between NH2 teams, as represented in Fig. 3b. Additionally, the broad peak of 3410 cm-1 confirms the in situ cross-linked reactions take positioned in PVC/LLDPE)ZnO-EDA pattern. Determine 4c exhibits the attribute peaks of aniline molecules in (PVC/LLDPE) ZnO-An pattern. The 2 FTIR peaks positioned at 3438 cm-1 and 1267 cm-1 correspond to the stretching vibration of (N–H) and (C–N) bonds in secondary amines as a result of response between aniline and PVC molecules in (PVC/LLDPE) ZnO-An pattern. Determine 4d exhibits the attribute FTIR peaks of (PVC/LLDPE)ZnO-pA positioned at 1609 cm-1 and 1047 cm-1 that project to the stretching and bending vibration of C–O and N–H bonds in p-anisidine molecules. Determine 4e present the attribute FTIR peaks of (PVC/LLDPE) ZnO -DMA positioned at 1223 cm-1 that project to the stretching vibration of C-N of tertiary amine and 1612 cm-1 that that project to the stretching vibration of C=C of a benzene ring with no NH peak noticed for dimethyl aniline molecules.
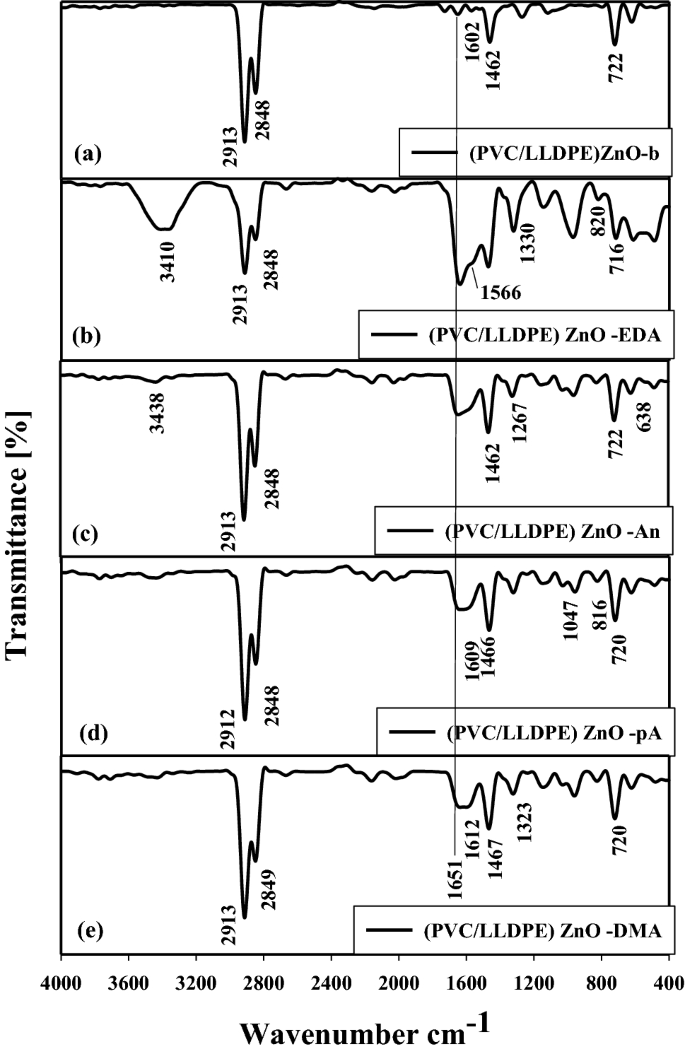
The FTIR/ATR curves for amined modified PVC in (PVC/LLDPE).
The XRD evaluation of modified (PVC/LLDPE)ZnO
Determine 5a exhibits the XRD sample of virgin LLDPE within the clean pattern of (PVC/LLDPE)ZnO-b has appeared at 20.51° and 23.18°, that are assigned to the 110 and 200 reflections of LLDPE. On the identical time, the XRD sample of virgin PVC movie at 2θ ~ 17° and 26° doesn’t seem. This could possibly be as a result of gamma irradiation which will trigger dechlorination (–HCl) of PVC molecules42. The addition of ZnO nanoparticles into (PVC/LLDPE) matrix offered new XRD peaks positioned at 30.86°, 34.4°, 36.3°, 47.51°, 56.61°, 62.81°, 66.41°, 67.91°, 69.10°, 72.52°, 76.9° that listed to ZnO sample of (100), (002), (101), (102), (110), (103), (200), (112), (201), (004) and (202). Determine 5b–e exhibits the XRD sample of (PVC/LLDPE)ZnO-b modified chemically by the nucleophilic substitution course of. In comparison with Fig. 1a, the intensities of the diffraction peaks considerably modified after chemical modification. Determine 3b exhibits the XRD peaks of (PVC/LLDPE) ZnO –EDA pattern that just about exhibit solely two peaks at 7.02° and 17.3°. Determine 5c exhibits the XRD peaks of (PVC/LLDPE)ZnO-An, which present the sharp attribute peaks of aniline molecules with excessive crystallinity at 16.38°29. Determine 5c,d exhibits the XRD peaks of each (PVC/LLDPE)ZnO–pA, (PVC/LLDPE)ZnO-DMA that exhibit the shifted peaks of LLDPE and ZnO sample.
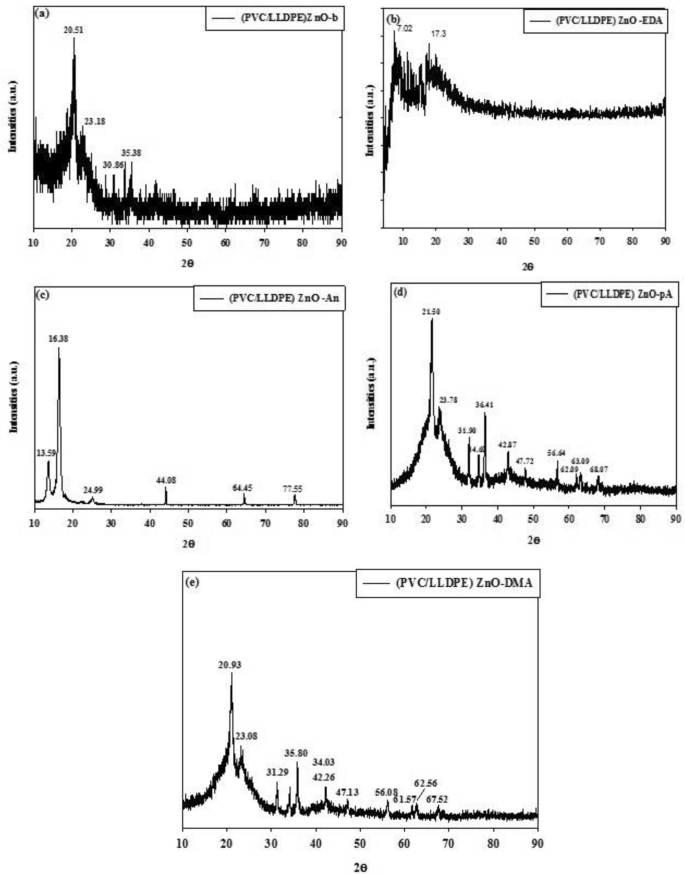
The XRD curves of aminated mopdfied PVC in (PVC/LLDPE)ZnO samples.
Thermal properties of modified (PVC/LLDPE)ZnO carried out by DSC
Differential scanning calorimetry was carried out to find out the melting transition temperature (Tm) and glass transition temperature (Tg) of (PVC/LLDPE)ZnO-b recycled mix and their chemical modification matrices. The 2 temperature is an important parameter in polymer characterization to judge the compatibilizing agent impact of SBR added to (PVC/LLDPE)ZnO-b pattern in addition to the chemical modification. Determine 6 exhibits the measurement DSC of modified (PVC/LLDPE)ZnO samples, Examine to the clean pattern, the 4 DSC curves of modified samples exhibited adjustments within the melting level of PVC. The obtained outcomes is confirmed that the amination reactions happen in PVC molecules by dechlorination. Determine 6a exhibit three melting factors (Tm) of irradiated (PVC/LLDPE)ZnO-b pattern, two melting factors for LLDPE on the temperature of 117 °C, 120 °C and 294 °C project to PVC molecules. The melting peak of LLDPE into double peaks positioned at 117 °C, 120 °C of the irradiated samples is as a result of presence of each irradiated and non-irradiated areas that give completely different melting temperatures. The very best melting level (Tm) at 120 °C is because of gamma irradiation-induced cross-linked reactions of LLDPE. As anticipated, the glass transition temperature (Tg) of (PVC) at ~ 85–93 °C disappeared. This could possibly be as a result of impact of compatibilizing agent (SBR) as a plasticizer, limiting the glassy temperature of PVC43. Determine 6b exhibits the DSC curve of (PVC/LLDPE) ZnO –EDA pattern that exhibited a peak overseas at 82 °C, akin to the moisture content material. The outcomes indicated that amination reactions by EDA is an efficient technique to extend the hydrophilicity of PVC. As well as, the elevated melting level (310 °C) of (PVC/LLDPE) ZnO–EDA pattern is as a result of intramolecular H bonds established after chemical modification as confirmed by FTIR knowledge. Determine 4 c-d exhibit the DSC of the three ((PVC/LLDPE)ZnO-An, (PVC/LLDPE) ZnO-pA and (PVC/LLDPE)ZnO-DMA)) samples with single Tg on the temperature of (98, 88 and 95) °C, respectively. PVC has Tg at a temperature of 93.5 °C in line with the DSC knowledge discovered within the literature44,45,46. This reality reinforces the plasticizing impact induced by p-anisidine, particularly the p.c of 5 w% of ZnO it might improve the Tg when it acts as a filler47,48. Moreover, when aniline and N, N, dimethyl aniline had been reacted with PVC molecules precipitated a rise of their Tg as a result of restriction on the free rotation and therefore restricted segmental movement49. Alternatively, the melting level (Tm) of PVC was decreased on the temperature of 257 °C, 251 °C and 277°C for ((PVC/LLDPE)ZnO-An, (PVC/LLDPE) ZnO-pA and (PVC/LLDPE)ZnO-DMA)) samples, respectively. The DSC knowledge confirmed the chemical amination modification of (PVC/LLDPE)ZnO-b samples.
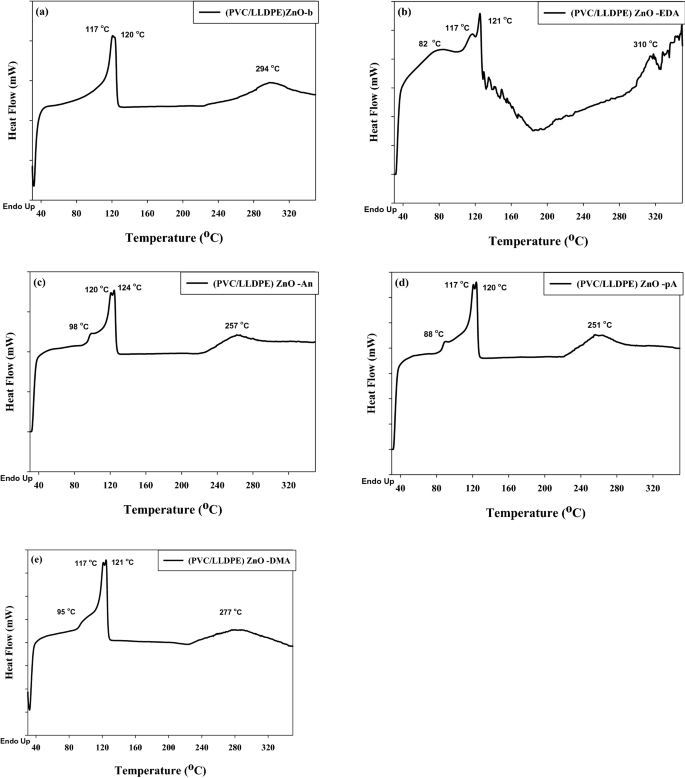
The DSC thermograms of aminated modified PVC in (PVC/LLDPE)ZnO.
The position of the aminated modified PVC/LLDPE on the oil elimination capability
Among the many aminating teams that had been used, aniline (An), p-anisidine and dimethyl aniline (DMA) seem to own the very best motor oil elimination capability (41, 38 and 43) %, respectively. The obtained knowledge confirmed that the three aminating reagents (An, pA and DMA) turn into extra motor oil elimination efficient than the clean pattern as a result of p.c of the fragrant benzene ring. The 2 samples of (PVC/LLDPE)ZnO-b and (PVC/LLDPE)ZnO-EDA appear to be the least efficient for this job as a result of absence of fragrant benzene ring and further NH2 group in (PVC/LLDPE)ZnO-EDA pattern. A lower in elimination capability of motor oil than castor oil was noticed for all samples as a result of adsorption of castor oil on amine teams as a result of formation of intermolecular H bonds. As proven in Fig. 7a the castor oil elimination capability of the clean pattern (PVC/LLDPE)ZnO-b modified by three amine reagents (EDA, An and DMA) was enhanced. (PVC/LLDPE)ZnO-EDA pattern and two different aminated modified samples of (PVC/LLDPE)ZnO-An and (PVC/LLDPE)ZnO-DMA confirmed excessive elimination capability of castor oil (53, 51 and 43) %, respectively. The structural knowledge of castor oil and aminated modified (PVC/LLDPE)ZnO-EDA pattern show the excessive adsorption capability as a result of intermolecular H-bonds established between C=O and OH teams in castor oils with NH2 teams in (PVC/LLDPE)ZnO-EDA as summarized in Fig. 7b. All of those modified structural parameters appear to be influenced by oil adsorption capability. This highlights that the distribution of amine websites on the adsorbents is important to excessive castor oil adsorption efficiency. Determine 7c highlights that the fragrant benzene ring in (PVC/LLDPE)ZnO-DMA provides hydrophobic websites on the floor of the adsorbent is important to the excessive efficiency of motor oil adsorption and water repellent.
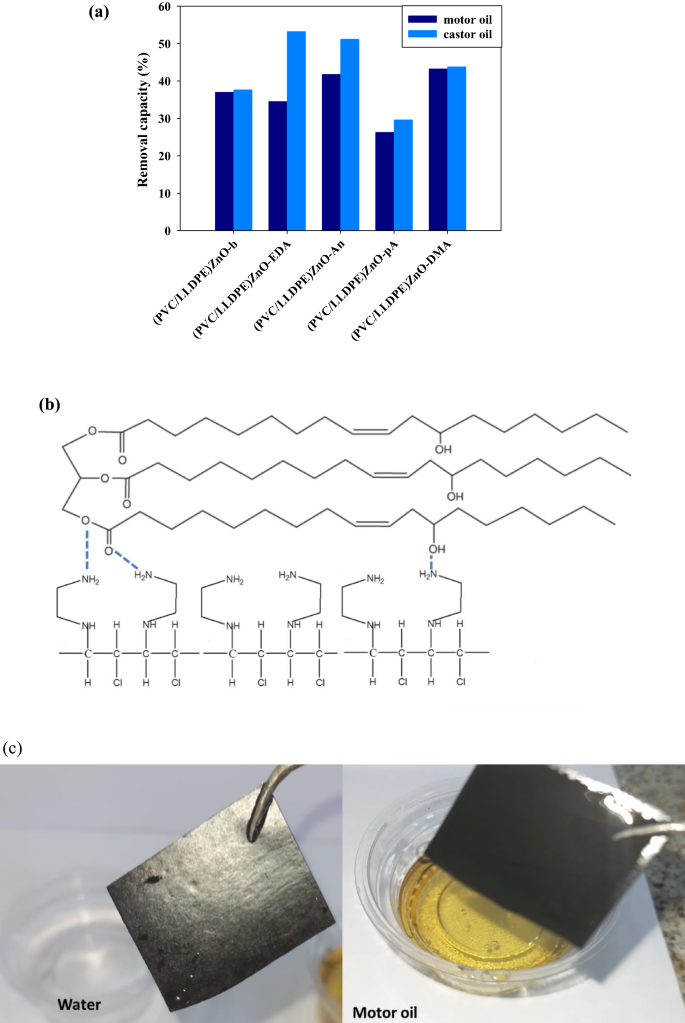
(a) adsorption capability of motor oil and castor oil by aminated modified PVC in (PVC/LLDPE)ZnO relying on the character of the aminating reagent, (b) the proposed intermolecular H-bonds formation between Castro oil and NH2 teams in (PVC/LLDPE)ZnO-EDA samples and (c) the effectivity of motor oil elimination by (PVC/LLDPE)ZnO-DMA.
The electrical conductivity of aminated modified PVC
Determine 8a represents {the electrical} conductivity irradiated clean (PVC/LLDPE)ZnO-b and modified aminated pattern. It’s noticed that the (PVC/LLDPE)ZnO-pA mix had greater electrical conductivity than different samples at any given frequency regardless of the elevated electrical conductivity of the PVC with the rise in frequency. The rise within the modfied mix’s conductivity could possibly be attributed to the formation of amine websites which can be within the protonation state. The protonation is chemically fashioned intermolecular and intramolecular H-bonds as an inorganic doping course of: the protonated of amine teams is understood to be extra conducting attributable to a excessive diploma of conjugation. The permittivity ε’ and dielectric loss ε” for aminated modified PVC samples had been represented in Fig. 8b,c over a frequency vary 0.01 Hz as much as 600 Hz. The measurements had been carried out at room temperature (25 ± 1 °C). From Fig. 8b,c, the values of ε’ lower by rising the utilized frequency exhibiting an anomalous dispersion. In such a spread, the permittivity has a contribution from orientation polarization. Additionally, ε’ & ε” improve so as (PVC/LLDPE)ZnO-pA, (PVC/LLDPE)ZnO-EDA, (PVC/LLDPE)ZnO-DMA, (PVC/LLDPE)ZnO-An and (PVC/LLDPE)ZnO-b. This improve in ε’ & ε” with the incorporation amino teams is as a result of rise in dipoles-dipoles interactions and intramolecular H bonds which ends up in a rise within the orientation polarization and likewise to the presence of interfacial polarization. The mix plastic used to fabricate medium voltage cables has many benefits like a low dissipation issue of about 0.03% at 20 °C, a low dielectric fixed of two.2–2.5, good thermo-mechanical properties and a excessive working temperature of about 90 °C50. The aged samples present rising dielectric fixed and dielectric loss51. In our samples, dielectric loss is extra minor for neat samples (PVC/LLDPE)ZnO-b. After chemical modification, the dielectric loss is elevated with out the ageing course of. It signifies that the existence of nanoparticles52 and amine websites on the pattern mix will improve dielectric loss. Subsequently it’s attainable to postulate that chemical modification will improve the dielectric fixed and dielectric loss.
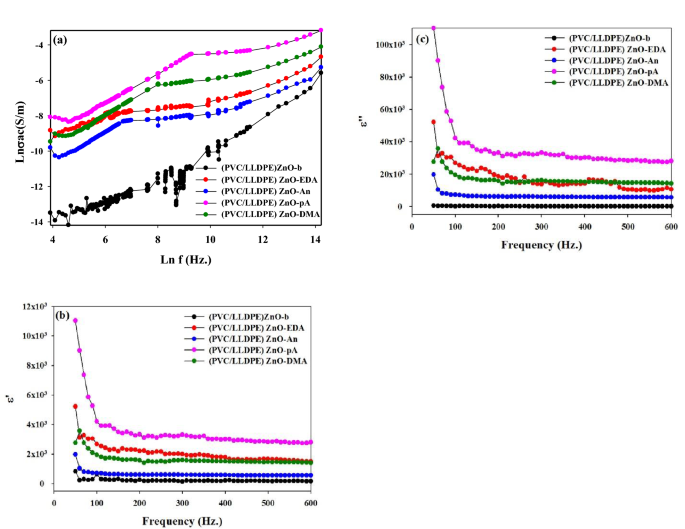
(a) The AC electrical conductivity, (b) permittivity εʹ and (c) the dielectric loss εʺ at room temperature ~ 25 °C of aminated modified PVC as a operate of frequency.
Simulation and modeling of electrical area distribution of aminated modified PVC
The cable was a single core 22 kV shielded underground medium voltage cable. COMSOL Multiphysics was used to simulate the electrical area distribution in Medium Voltage Cables on this research. The electrical area distribution has been studied, ranging from the copper core to the outer semiconductor layer of the cable. Determine 9a exhibits that at 1 mm of arc size, the distribution of the electrical area inside (PVC/LLDPE)ZnO-b pattern marked cables is non-uniform. For (PVC/LLDPE)ZnO-pA pattern, as proven in Fig. 9b the electrical area distribution is beginning to turn into uniform and step by step decreases from the within to the skin. It’s because the p-anisidine stuffed (PVC/LLDPE)ZnO-pA pattern preserves a uniform electrical area and reduces electrostatic rigidity, rising relative permittivity values for internal semiconductors and outer semiconductors from 2.05 to 2.23. The optimum AC conductivity (AC: 2.44 × 10–4 S/m) in minimal relative permittivity (2.23) was achieved for (PVC/LLDPE)ZnO-pA irradiated at 20 kGy. Determine 9c,d display the electrical potential distribution of (PVC/LLDPE)ZnO-b and (PVC/LLDPE)ZnO-pA samples, respectively with no change famous within the conduct curve. The electrical potential distribution step by step decreases for 2 samples from 22,000 to 0 V.
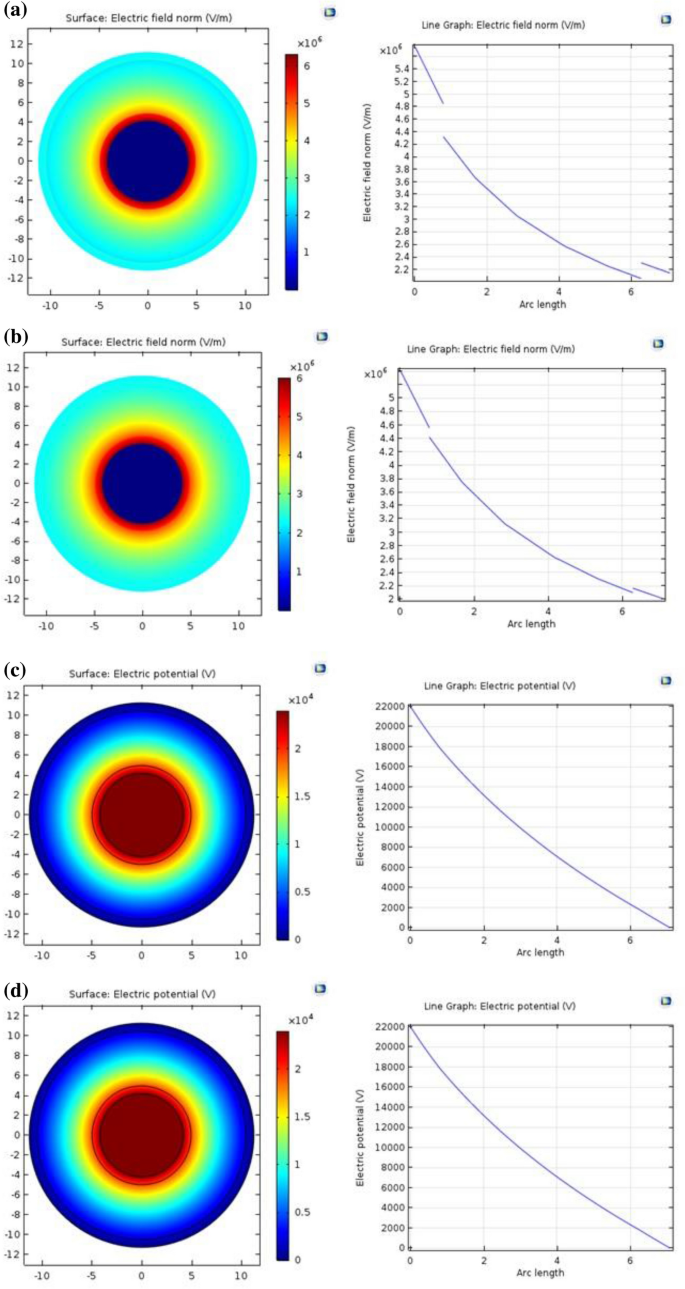
Electrical area distribution in medium-voltage cable of (a) (PVC/LLDPE)ZnO-b. (b) (PVC/LLDPE)ZnO-pA. (c) (PVC/LLDPE)ZnO-b. (d) (PVC/LLDPE)ZnO-pA pattern.
[ad_2]
Supply hyperlink